 
Therefore, there remains a need for effective biomarkers and potential targets to improve the diagnosis and treatment of patients with brain metastasis.Īccumulating evidence, including our work on multi-omics subtyping analysis, suggests that the tumor environment (TME) plays a major role in brain metastasis patients’ responses to stereotactic radiosurgery and whole-brain radiation therapy 9. Unfortunately, the mortality rate and the recurrence of brain metastasis remain high 8. Substantial advances have been made in the diagnosis and treatment of patients with metastatic brain tumors, including targeted therapies that penetrate blood brain barrier and stereotactic radiosurgery as primary strategies of therapy 7. Brain metastasis are 10 times more common than primary brain tumors 6 and is associated with poor survival outcomes 6. The majority of brain metastases are from primary cancers such as lung, breast, and melanoma 5. It is estimated that brain metastasis occurs in at least 6% of all newly diagnosed cancer cases 1, 2, 3, and is a devastating cancer complication for the 200,000 patients each year diagnosed in the US 4. These discoveries provide potential biomarkers for effective therapeutic targets and intervention strategies.īrain metastasis refers to the malignant tumors that metastasize to the brain. Taken together, our comprehensive analyses identify type I collagen-secreting tumor-associated fibroblasts as key mediators in metastatic brain tumors and uncover tumor receptors that are potentially associated with patient survival. 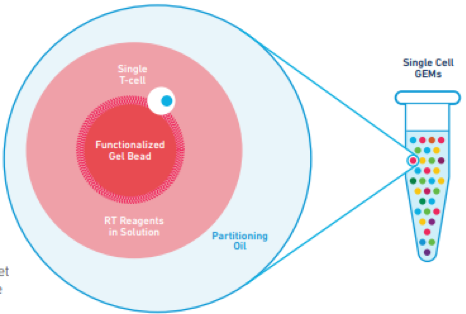
Furthermore, tumor cell-specific receptors exhibit a significant association with patient survival in both brain metastasis and native glioblastoma cases. 
Additionally, we observe M1 activation in native microglial cells and infiltrated macrophages, which may contribute to a proinflammatory TME and the upregulation of collagen type I expression in fibroblasts. These fibroblasts exhibit high expression of type I collagen genes, dominate cell-cell interactions within the TME via the type I collagen signaling axis, and facilitate the remodeling of the TME to a collagen-I-rich extracellular matrix similar to the original TME at primary sites. Importantly, we identify tumor-associated fibroblasts in both our in-house dataset and external scRNA-seq datasets. Interestingly, distinct cellular compositions are observed across different samples, indicating the influence of diverse cellular interactions on the infiltration patterns within the TME. Our analysis reveals the presence of various intratumoral components, including tumor cells, fibroblasts, myeloid cells, stromal cells expressing neural stem cell markers, as well as minor populations of oligodendrocytes and T cells. In this study, we have applied scRNA-seq and profiled 10,896 cells collected from five brain tumor tissue samples originating from breast and lung cancers. Recently, the remarkable breakthroughs in single-cell RNA-sequencing (scRNA-seq) technology have advanced our insights into the tumor microenvironment (TME) at single-cell resolution, which offers the potential to unravel the metastasis-related cellular crosstalk and provides the potential for improving therapeutic effects mediated by multifaceted cellular interactions within TME. Therefore, there is a critical need to identify effective biomarkers that can support frequent surveillance and promote efficient drug guidance in brain metastasis. 
It is a leading cause of death in advanced-stage cancer, resulting in a five-year overall survival rate below 10%. Brain metastases is the most common intracranial tumor and account for approximately 20% of all systematic cancer cases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
